Owing to our business policies, client centric approach and competitive price structure, we have been continuously expanding our list of precious customers. We offer these products in several packagings to meet the accurate needs of our precious customers. The offered products are highly demanded for their features like long shelf life, accurate composition, moisture proof packaging, etc. These products are manufactured under the supervision of experienced professionals using the best quality ingredients and modern technology. We are also engaged in offering a superior quality range of Nickel Sulphate, Electroplating Chemical, Nickel Metallic Salt, Specialty Chemical, Chromic Acid Flake, Copper Metalic Salt, Copper Sulphate, Chromium Metallic Salt, Zinc Metallic Salt, Tin Metallic Salt, Cobalt Metallic Salt, Cadmium Metallic Salt, Sodium Salt, Silver Salt, Potassium Dichromate, Metal Finishing Chemical and Cyanides Chemical. This is a complex with 6 water ligands surrounding the central metal ion. This is due to the acidic nature of Cr3+ (aq). Explanation: You might assume that you would get a precipitate of chromium (III) carbonate, but this is not the case. 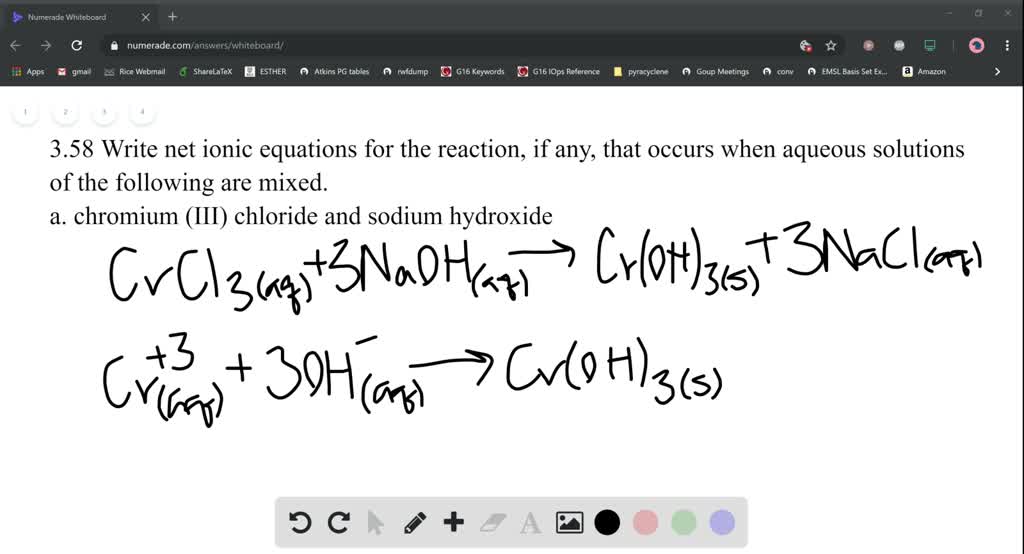
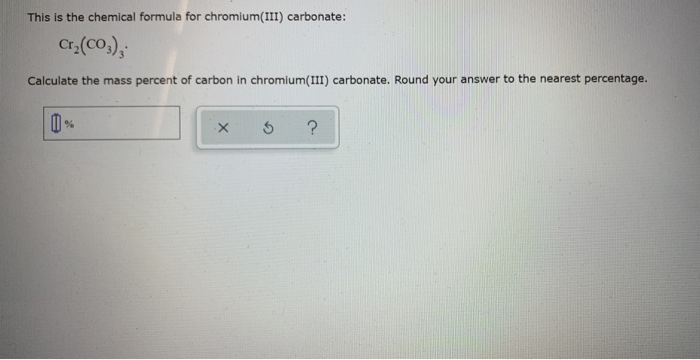
Advertisement AkshayG The name of the compound is. This compound is composed of 36.6 Chromium, 12.7 Carbon, and 50.7 Oxygen. The formula of chromium (III) carbonate is (Cr (CO)) Let us calculate the molar mass of (Cr (CO)) molar mass of (Cr (CO)) 2X atomic mass of Cr + 3X atomic mass of C + 9X atomic mass of O molar mass of (Cr (CO)) 2X 52 + 3X12 +9X16 284g/mol As per the formula there are nine moles of oxygen in each mole of (Cr (CO)). Chromium(III) chloride anhydrous - Physico-chemical Properties Molecular Formula, Cl3Cr Molar Mass, 158.36 Density, 2.87g/mLat 25C(lit.) Melting Point. Chromium has 2 atoms, Carbon has 3 atoms, and Oxygen has 9 atoms. 
It is composed of Chromium, Carbon, and Oxygen. Since it carries a #color(blue)(3+)# charge, it follows that you must use the (III) Roman numeral.Established in the year 2007, at Ahmedabad (India), we “Mahavir Meta Chem” are known as the notable manufacturer and supplier of a broad assortment of Electroplating Chemicals and Metallic Salts. You get a precipitate of chromium (III) hydroxide and CO2 gas. Cr2 (CO3)3 is Chromium (III) Carbonate or Chromic Carbonate. EINECS 249-781-1 Chromic carbonate Carbonic acid,chromium salt Chromium carbonate Basic chromium. #color(red)(2) xx "Cr"^(color(blue)(3+)" "# and #" " color(blue)(3) xx "CO"_3^(color(red)(2-)#īecause chromium is a transition metal, which means that it can have multiple oxidation states, you must use Roman numerals to express its oxidation state in the compound. This means that the ions that make up this compound are This means that the charge on the cation will be equal to #color(blue)(3+)#. Chromium(III) acetylacetonate Chromium(III) basic sulfate Chromium(III) bromide Chromium(III) carbonate hydrate Chromium(III) chloride Chromium(III). Notice that it's written between parentheses, which tells you that it contains one atom of carbon and three atoms of oxygen, and that it has a subscript of #color(blue)(3)#. Now, the anion is actually a polyatomic ion called the carbonate anion. It has several different forms, the most common of. Since it has a subscript of #color(red)(2)#, it follows that the charge of the anion must be equal to #color(red)(2-)#. Chromium Carbonate is an odorless, hard, steel-gray, lustrous metal available in crystals or powder. Get the Chromium III Nitrate Formula along with its properties, chemical structure, and uses. decarboxylation of two carbonate-bridged complexes of chromium(III) and. Chromium III nitrate is a group of inorganic compounds that are comprised of chromium, nitrates with varying amount of water. In your case, the compound contains chromium, #"Cr"#, as its cation. complexation of chromium(III) to NOM was intermediate between that of iron(III). This is because chromium (Cr) is a transition metal and transition metals can have. C 2235 (OTTO) Chromium carbonate Cas 2 - used in electrophoretic. No we cannot write chromium(III) carbonate as chromium carbonate. It's important to remember that ionic formulas are written using the crisscross rule, which states that when a cation and an anion form an ionic compound, the charge on the cation becomes the subscript of the anion and the charge of the anion becomes the subscript of the cation in the chemical formula of the compound. C 2235 (OTTO) Chromium carbonate - 2 - is an inorganic compound. #"Cr"_ color(red)(2) ("CO"_ 3)_color(blue)(3)# Chromium Carbonate is a water insoluble Chromium source that can easily be converted to other Chromium compounds, such as the oxide by heating (calcination). In order to name an ionic compound, you must identify the cation, which is the positively charged ion, and the anion, which is the negatively charged ion.Ĭations are always written first in the chemical formula of an ionic compound, followed by the anions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
